The Regulation framework for our Daysoft contact lenses.
Contact lenses are a medical device, and like all medical devices being sold on the European market, daysoft lenses must carry the CE Mark. This can be seen with the symbol ‘CE’ followed by a number, in our case, 1639, which represents the Notified Body that performs Audits on Daysoft on a regular basis. In our case our Notified Body is SGS. The chart below shows the process through which we need to pass to sell our lenses.
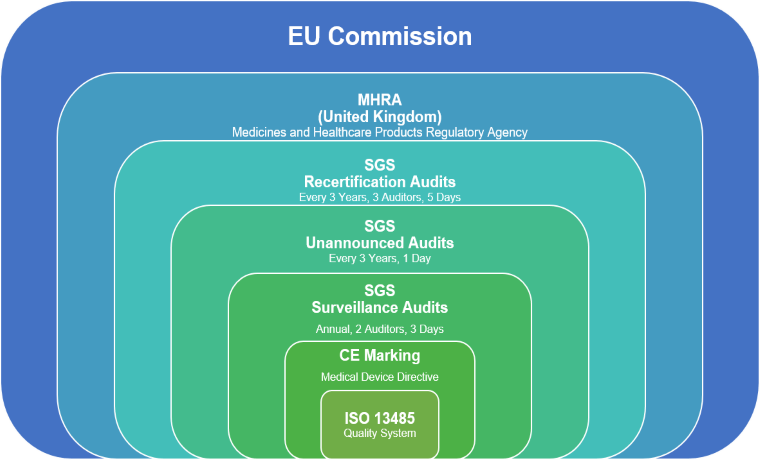
ISO 13485 Quality System
All daysoft lenses are manufactured under the control of a certified Quality Management System, compliant with BS EN ISO 13485. All processed in the manufacture of our lenses are completed under one roof at our plant in Scotland.
CE Marking
Additional requirements of the European Medical Devices Directive (93/42/EEC) have also been implemented, maintained and assessed to allow our lenses to be CE marked.
SGS Audits
As mentioned above, Daysoft is subject to Audits by SGS on a regular basis:
SGS Surveillance audits are carried out annually to ensure ongoing compliance to BS EN ISO 13485. Among the things checked in the audit is things like the effectiveness of the sterilisation process. This is obviously something that is critical for something that is going to be inserted into an eye.
Daysoft will be subject to an unannounced Audit by SGS a minimum of once every three years with absolutely no prior notice given.
Re-certification Audits are a more in depth audit which takes place every three years.
MHRA (Medical and Healthcare products Regulatory Agency)
Our Notified Body, SGS is also strictly governed and assessed by the MHRA which is an executive department of the Department of Health in the UK.
EU Commission
The MHRA operate under the guidelines and recommendations of the EU Commission.